CAR T-Cell Therapy
 Your immune system is naturally equipped to fight cancer, but cancer cells can learn to outmaneuver your body’s defense system, allowing them to grow and spread. An innovative type of immunotherapy called CAR T-cell therapy can revitalize the cancer-fighting ability of your immune system through your T cells by adding a gene that helps them locate and destroy cancer cells. As the first center offering CAR T-cell therapy in Kentucky, Markey Cancer Center provides this leading-edge treatment to patients from the Bluegrass and beyond who have certain blood cancers.
Your immune system is naturally equipped to fight cancer, but cancer cells can learn to outmaneuver your body’s defense system, allowing them to grow and spread. An innovative type of immunotherapy called CAR T-cell therapy can revitalize the cancer-fighting ability of your immune system through your T cells by adding a gene that helps them locate and destroy cancer cells. As the first center offering CAR T-cell therapy in Kentucky, Markey Cancer Center provides this leading-edge treatment to patients from the Bluegrass and beyond who have certain blood cancers.
Side effects of CAR T-cell therapy
Two major side effects may occur in the days and weeks following treatment, and symptoms may be mild or moderate. These side effects include:
- Cytokine release syndrome (CRS). CRS is widespread inflammation that may occur as the newly empowered immune system attacks cancer cells. This condition can cause a variety of symptoms, including fever, fatigue, a rapid heartbeat and difficulty breathing.
- Neurotoxicity. CAR T-cell infusion may cause problems with the nervous system. Symptoms of neurotoxicity may include confusion, dizziness, and difficulty speaking, concentrating or staying awake.
CRS and neurotoxicity are treatable and in the vast majority of cases, go away completely. Be sure to let your medical team know right away if you experience symptoms after returning home.
What is CAR T-cell therapy?
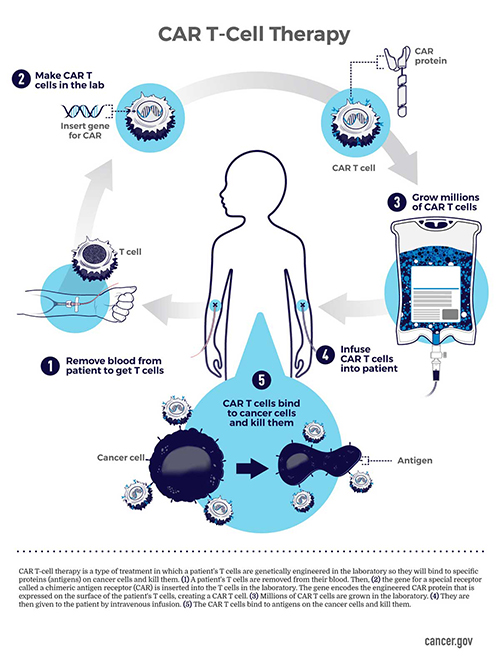
T cells or T-lymphocytes are a type of white blood cell (WBC) that can attack cancer. However, in some cases, cancer cells develop ways to avoid detection by T cells. That’s where CAR T-cell therapy comes in. This treatment adds a laboratory-created ingredient called a chimeric antigen receptor (CAR) to your T cells. The lab creates hundreds of millions of CAR T cells that, once reintroduced into your body, seek out and kill cancer cells.
The U.S. Food and Drug Administration approved the first CAR T-cell therapy in 2017. Since then, five more have received approval in the treatment of various blood cancers. Five approved CAR T-cell therapy products are available through Markey Cancer Center while the groundwork is being laid for the introduction of the latest approved therapy (FDA approval in February 2022).
That’s good news for patients with certain blood cancers. Before the introduction of CAR T-cell therapy, these patients had no other option if longstanding treatments, such as chemotherapy, proved ineffective. Now, CAR T-cell therapy gives these patients another chance for successful treatment. For some types of cancer, CAR T-cell therapy is quite effective.
Types of cancer treatable with CAR T-cell therapy
Currently, CAR T-cell therapy is available for patients with cancers that have not responded to other forms of treatment — known as refractory disease — or those who have relapsed. These types of cancer include:
- Acute lymphoblastic leukemia
- Diffuse large B-cell lymphoma
- Follicular lymphoma
- Mantle cell lymphoma
- Multiple myeloma
- Primary mediastinal B-cell lymphoma
- Transformed follicular lymphoma
What to expect during treatment
- First, you’ll be seen for a consultation by one of the hematologists practicing with the Hematology Program at Markey Cancer Center. They will evaluate your cancer to determine if CAR T cells are the best approach for you.
 You will then undergo testing to ensure you’re in the right physical shape for this treatment while also accessing numerous resources available through Markey for us to best support you through this treatment.
You will then undergo testing to ensure you’re in the right physical shape for this treatment while also accessing numerous resources available through Markey for us to best support you through this treatment.- Your T cells will then be collected (a process called apheresis). During this two- to three-hour process, your physicians will collect blood, remove T cells and return the blood to your body.
- Next, the center sends your T cells to a manufacturing lab. The lab modifies your cells by adding CAR, and then creates millions of CAR T cells. This process may take three to four weeks.
- While the lab prepares your CAR T cells, you may receive “bridging” chemotherapy to keep the cancer in check. The choice of giving bridging chemotherapy is individualized and not everyone may benefit from it.
- When the T cells are ready, you will receive a low dose of lymphodepleting chemotherapy prior to receiving the manufactured cells via intravenous infusion at Markey. This process takes about 20-30 minutes. Once in your body, the cells begin hunting and attacking cancer cells.
- You may stay in the hospital for a few days while a specialist CAR-T team consisting of physicians, nurse practitioners, nurses and pharmacists monitor you for side effects of treatment. All CAR-T patients are cared for on the 11th floor of UK Albert B. Chandler Hospital Pavilion A, which has been specially set up for patients receiving treatments for blood cancer.
- When you go home, you’ll watch for side effects. A few weeks into receiving CAR-T therapy, your Markey hematologist will assess how well your cancer has responded. After a minimum follow-up period, you’ll resume care with your primary hematologist/oncologist while continuing periodic visits with your CAR T-cell therapy program.
Why choose UK Markey Cancer Center?
CAR T-cell therapy is one of the most innovative types of cancer treatment, and Markey is at the forefront of U.S. centers offering this therapy. Here’s what sets our program apart:
- A comprehensive, patient-centered approach to care. At Markey, you’ll have access to a multidisciplinary team of physicians, nurses, pharmacists, social workers and CAR T-cell therapy coordinators. Ours is a diverse team but also small enough that we can offer patients personalized attention and all-encompassing care.
- Cellular therapy accreditation. Our program is accredited by the Foundation for the Accreditation of Cellular Therapy. That’s an indicator of excellence in cellular treatments, such as CAR T-cell therapy.
- Commitment to quality. Markey is Kentucky’s only National Cancer Institute-designated cancer center. That’s a testament to our commitment to high-quality, innovative care.
- Experience. Ours was the first CAR T-cell therapy program in Kentucky. We’ve treated dozens of patients since establishing the program in 2018.
Shaping the future of CAR T-cell therapy
Markey is part of a multicenter clinical trial studying the safety and effectiveness of off-the-shelf CAR T cells. This product uses donor cells modified with CAR instead of patients’ T cells. For eligible patients, using an off-the-shelf product eliminates the need to obtain their T cells and send them off to a lab to add CAR. That can reduce the time to treatment from weeks to a few days. In addition, using off-the-shelf CAR T cells eliminates the need for patients to get bridging chemotherapy before CAR T-cell therapy to keep cancer from progressing.